The gases can be used in both regular and bag-on-valve (BOV) aerosols. Mechanical break-up
(MBU) actuators must be used to achieve a spray, which is generally medium/coarse to coarse. With
other actuators, the product may simply stream out, going as much as 20' (6.10 meters) when the aerosol
dispenser is initially used. However, as the product is used, the pressure decreases and the “reach”
of the stream lessens. This will be important for wasp and hornet sprays, but less so for yellow jacket
bee sprays where the insects live in underground nests.
Nitrogen is almost insoluble
in water: 2.35mL per100 mL of
water at 32°F (0°C) and 760 mm
of Hg. The solubility increases
almost linearly with increasing
pressure, so that about 23.2mL
dissolves in 100mL of water
at 130 psi-g. at 32°F. About
16.2mL dissolves at 77°F (25°C).
Note: the volume of dissolved
nitrogen is illustrated for
standard pressure and temperature
(STP) conditions: 0°C and
760mm.
Nitrogen is much less soluble
than oxygen or air, both in water
and other liquids. For pressure
considerations, the tiny solubility
can be neglected. However,
it becomes physically evident
in the dispensing of foams and
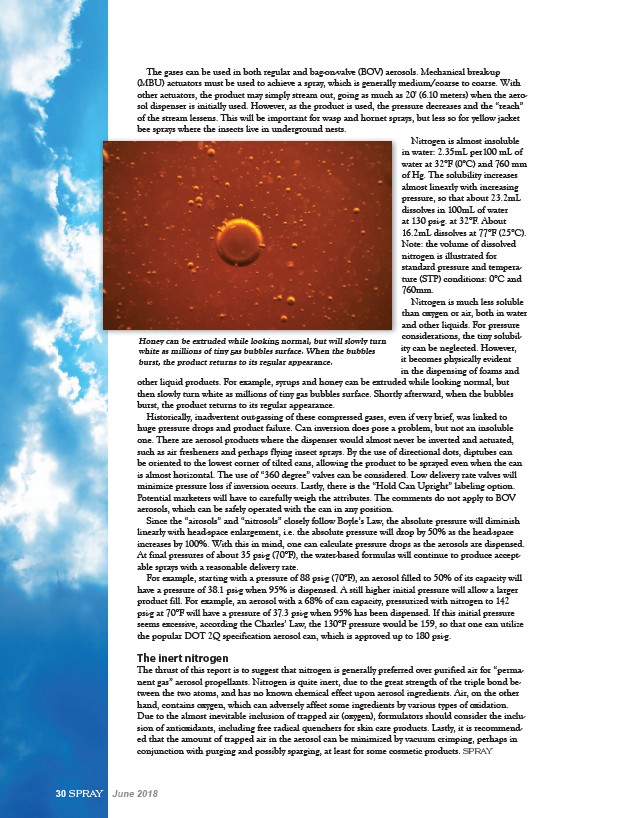
Honey can be extruded while looking normal, but will slowly turn
white as millions of tiny gas bubbles surface. When the bubbles
burst, the product returns to its regular appearance.
other liquid products. For example, syrups and honey can be extruded while looking normal, but
then slowly turn white as millions of tiny gas bubbles surface. Shortly afterward, when the bubbles
burst, the product returns to its regular appearance.
Historically, inadvertent out-gassing of these compressed gases, even if very brief, was linked to
huge pressure drops and product failure. Can inversion does pose a problem, but not an insoluble
one. There are aerosol products where the dispenser would almost never be inverted and actuated,
such as air fresheners and perhaps flying insect sprays. By the use of directional dots, diptubes can
be oriented to the lowest corner of tilted cans, allowing the product to be sprayed even when the can
is almost horizontal. The use of “360 degree” valves can be considered. Low delivery rate valves will
minimize pressure loss if inversion occurs. Lastly, there is the “Hold Can Upright” labeling option.
Potential marketers will have to carefully weigh the attributes. The comments do not apply to BOV
aerosols, which can be safely operated with the can in any position.
Since the “airosols” and “nitrosols” closely follow Boyle’s Law, the absolute pressure will diminish
linearly with head-space enlargement, i.e. the absolute pressure will drop by 50% as the head-space
increases by 100%. With this in mind, one can calculate pressure drops as the aerosols are dispensed.
At final pressures of about 35 psi-g (70°F), the water-based formulas will continue to produce acceptable
sprays with a reasonable delivery rate.
For example, starting with a pressure of 88 psi-g (70°F), an aerosol filled to 50% of its capacity will
have a pressure of 38.1 psi-g when 95% is dispensed. A still higher initial pressure will allow a larger
product fill. For example, an aerosol with a 68% of can capacity, pressurized with nitrogen to 142
psi-g at 70°F will have a pressure of 37.3 psi-g when 95% has been dispensed. If this initial pressure
seems excessive, according the Charles’ Law, the 130°F pressure would be 159, so that one can utilize
the popular DOT 2Q specification aerosol can, which is approved up to 180 psi-g.
The inert nitrogen
The thrust of this report is to suggest that nitrogen is generally preferred over purified air for “permanent
gas” aerosol propellants. Nitrogen is quite inert, due to the great strength of the triple bond between
the two atoms, and has no known chemical effect upon aerosol ingredients. Air, on the other
hand, contains oxygen, which can adversely affect some ingredients by various types of oxidation.
Due to the almost inevitable inclusion of trapped air (oxygen), formulators should consider the inclusion
of antioxidants, including free radical quenchers for skin care products. Lastly, it is recommended
that the amount of trapped air in the aerosol can be minimized by vacuum crimping, perhaps in
conjunction with purging and possibly sparging, at least for some cosmetic products. Spray
30 Spray June 2018